Abstract
Background:
A substantial percentage of patients transfused with blood products develop HLA allo-immunization. Although HLA-matched platelets are effective in treating thrombocytopenia for those with HLA antibodies, a significant proportion of patients lack HLA-compatible platelet products resulting in platelet transfusion refractoriness and life-threatening thrombocytopenia. We and others have shown that compliment activation leading to the destruction of platelets bound by HLA allo-antibodies may play a pathophysiologic role in platelet refractoriness. Eculizumab is a monoclonal antibody that binds and inhibits C5 complement, blocking both the classic and alternative pathways of complement. Here we investigated whether treatment with eculizumab could be used as a treatment in HLA allo-immunized patients to overcome platelet transfusion refractoriness.
Methods:
We conducted a phase II pilot trial (NCT02298933) of eculizumab treatment in 10 subjects who had 1) HLA allo-immunization (detectable HLA class I antibodies), 2) severe thrombocytopenia and 3) platelet transfusion refractoriness. Patients were treated with a single infusion of eculizumab at a dose of 1200mg. Platelet refractoriness was defined as a transfusion corrected platelet count increment (CCI) of <7500/ul and <5000/ul 10-60 minutes and 18-24 hours respectively after platelets were transfused on at least 2 consecutive transfusions. Patients were deemed to respond to therapy if one of the first 2 platelet transfusions following eculizumab treatment resulted in a CCI>7500/uL at 10-60 minutes together with a CCI>5000/uL 18-24 hours post transfusion. Subjects were taken off study 14 days following eculizumab treatment. Responding patients who developed recurrent platelet refractoriness were eligible to re-enroll on study. All patients were required to receive meningococcal vaccination before eculizumab administration +/- antibiotic prophylaxis to cover N. meningitidis.
Results:
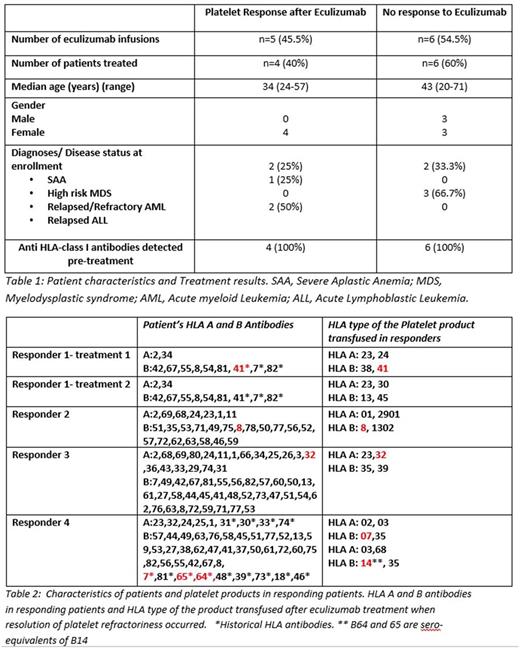
As of 4/2017, 11 Eculizumab infusions were administered to 10 HLA allo-immunized subjects (median age 39.5 years, range 20-71) with severe thrombocytopenia associated with platelet transfusion refractoriness. Patients had a diagnosis of SAA (n=4), refractory/relapsed AML (n=3); relapsed ALL (n=2) and high risk MDS (n=1).
Total complement (CH50) was the most reliable measurement for complement inhibition; CH50 decreased to <10U/ml in all treated patients. Four of 10 (40%) patients had a response to therapy with platelet refractoriness resolving following 5/11 (45.5%) eculizumab administrations (Table1). The estimated response rate for the 10 treated subjects was 40%, with a 95% confidence interval [CI] (17%-69%) and a standard deviation [SD] =0.155. Subject 1 received a 2nd treatment with eculizumab 2 months after her 1st response and again had resolution of platelet refractoriness.
Remarkably, platelet refractoriness was overcome with eculizumab treatment in patients receiving HLA incompatible platelets; in 4 out of 5 cases where platelet refractoriness was overcome, the administered platelet product given immediately after eculizumab treatment expressed an HLA allele for which an HLA antibody had been detected in the patient's serum (Table 2) . Resolution of platelet refractoriness resulted in a clinically meaningful reduction in the requirement for platelet transfusions: the median number of platelet transfusion given 2 weeks before and 2 weeks after the eculizumab infusion in responding patients was 9 (range from 3 to 11) and 4 (range from 3 to 5) transfusions respectively. For the non-responders, the median number of platelet transfusions given 2 weeks before and 2 weeks after the eculizumab infusion was 8 (range from 5 to 10) and 9.5 (range from 5 to 15) transfusions respectively.
Conclusions:
Final data from this pilot trial suggest eculizumab has efficacy in overcoming HLA antibody-associated platelet transfusion refractoriness. Importantly, these data reveal that eculizumab has the ability to overcome platelet transfusion refractoriness in patients with broad HLA allo-immunization in situations where HLA compatible platelets are unavailable. This study establishes proof of principle of the therapeutic role that compliment inhibition can play in overcoming platelet transfusion refractoriness and lays the foundation for a larger multicenter trial to define the overall efficacy of this approach.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.